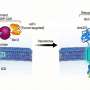
Researchers have made significant advancements in cancer therapy by developing remote-controlled chimeric antigen-receptor (CAR) T cells that utilize the drug venetoclax. This innovative approach aims to enhance the effectiveness of CAR-T cells, particularly in targeting solid tumors, which account for the majority of cancer cases.
CAR-T cells have already shown promise in treating blood cancers, but their efficacy against solid tumors has been limited. These engineered immune cells can unintentionally harm healthy cells, leading to serious side effects or even fatal immune overreactions. The introduction of venetoclax is intended to address these challenges by providing a mechanism to control CAR-T cell activity more precisely.
Potential Breakthrough in Cancer Treatment
The integration of venetoclax into CAR-T cell therapy represents a potential breakthrough in oncology. Venetoclax is a drug that inhibits a specific protein involved in cell survival, which can help to disengage CAR-T cells from tumors when necessary. This capability is particularly crucial for minimizing damage to healthy tissues surrounding solid tumors.
Researchers have conducted preliminary studies indicating that these modified CAR-T cells can effectively target cancer cells while reducing the risk of collateral damage. By controlling the activity of these immune cells, clinicians may be able to improve patient safety and treatment outcomes, particularly for those suffering from hard-to-treat solid tumors.
Addressing Limitations of Current Therapies
The challenges facing CAR-T cell therapies are significant. While they have revolutionized treatment options for certain blood cancers, their limitations in addressing solid tumors have left a gap in cancer treatment strategies. According to studies, solid tumors often present an environment that impedes the effectiveness of CAR-T cells, resulting in lower response rates compared to hematologic malignancies.
In addition, the potential for CAR-T cells to cause serious side effects remains a pressing concern. The systemic immune overreaction can lead to life-threatening conditions, complicating the therapeutic landscape. The use of venetoclax aims to mitigate these issues by allowing physicians to control the timing and intensity of CAR-T cell activation.
As research progresses, the implications of this technology could reshape the future of cancer therapy. The ability to remotely control CAR-T cells could not only enhance their efficacy against various cancer types but also provide a safer treatment option for patients.
In conclusion, the development of remote-controlled CAR-T cells using venetoclax marks a promising step forward in cancer treatment. Continued research and clinical trials will be essential to determine the full potential of this innovative therapy and its impact on patient care.