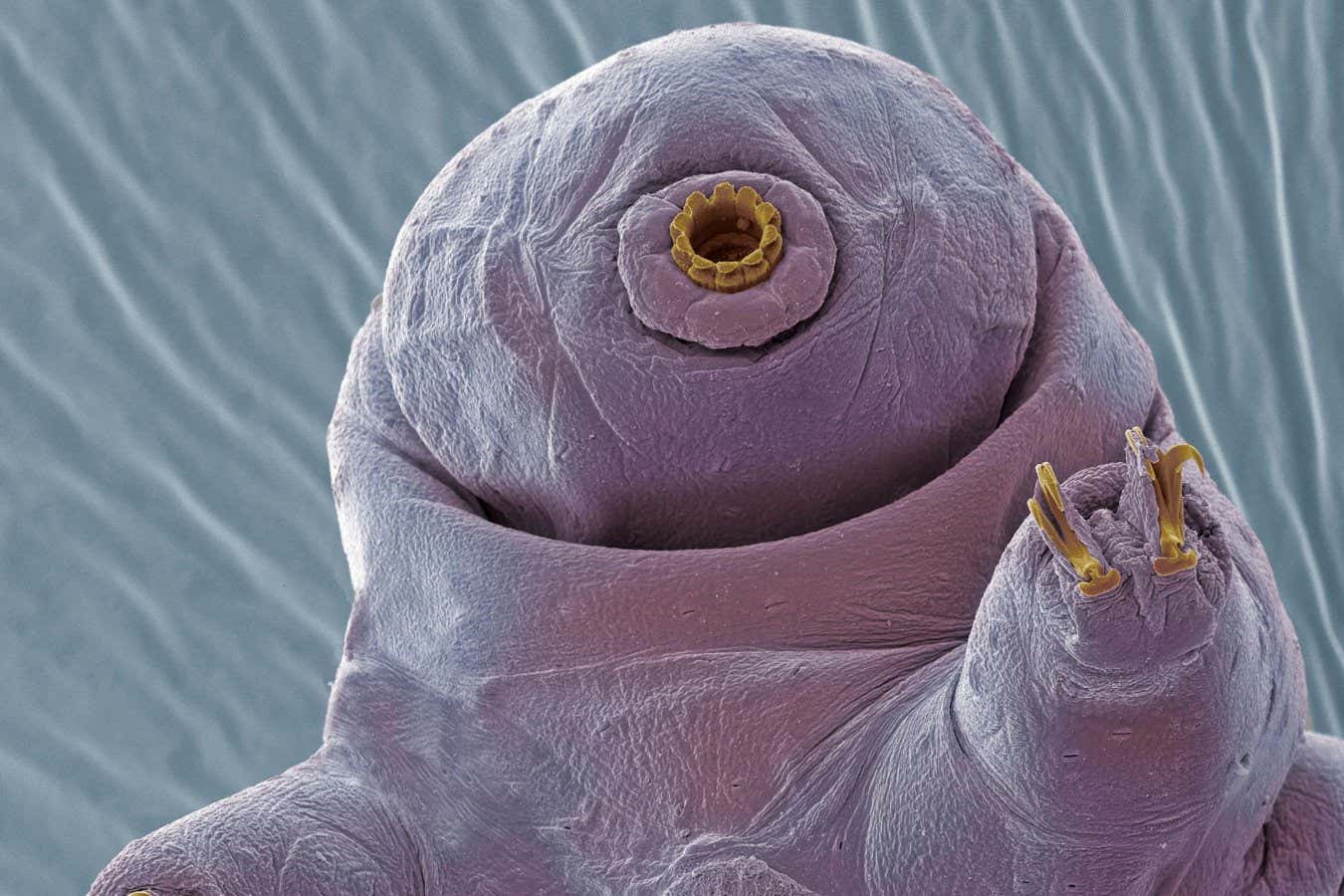
Scientists have encountered unexpected challenges in utilizing the resilient properties of tardigrades to protect astronauts from the harsh conditions of space. Tardigrades, often referred to as “water bears,” are renowned for their remarkable ability to survive extreme environments, including intense radiation and the vacuum of space. A team led by Corey Nislow at the University of British Columbia in Vancouver has investigated a specific protein from these micro-animals, known as Dsup, or damage suppressor. While Dsup shows promise in shielding cells from radiation and other mutagenic chemicals, its application poses significant limitations.
The research reveals that although Dsup can protect against a broader range of harmful agents than previously understood, it comes at a cost. According to Nislow, “There’s a cost for every benefit that we’ve seen.” High concentrations of Dsup were found to be fatal to yeast cells, while even moderate levels impaired cell growth. This presents a dilemma for future applications, as achieving a balance between protection and cell viability remains complex.
Tardigrades possess unique biological mechanisms that enable them to endure extreme stressors. In a notable study conducted in 2016, scientists discovered that Dsup plays a critical role in this resilience. When human cells were genetically engineered to produce Dsup, they exhibited increased resistance to radiation without adverse effects. This finding led to the hypothesis that Dsup could serve as a protective measure for astronauts facing cosmic radiation during long-duration space missions.
One proposed method for delivering Dsup involves using mRNA technology encased in lipid nanoparticles (LNPs), similar to the approach employed in mRNA COVID-19 vaccines. Nislow initially supported this strategy, believing that it could effectively provide astronauts with a protective countermeasure against DNA damage.
Despite the initial enthusiasm, Nislow’s team conducted extensive studies on yeast cells modified to produce Dsup. The results indicated that high levels of the protein proved detrimental, while lower levels still hindered cell growth. Dsup appears to safeguard DNA by physically enveloping it, complicating access for proteins responsible for DNA replication and repair.
As the research progresses, experts are considering how Dsup might be utilized to protect not only humans but also animals and plants in space. Nislow emphasizes the necessity of ensuring that Dsup is produced at optimal levels in specific cells where it is needed. James Byrne from the University of Iowa echoes this view, noting that continuous production of Dsup throughout the body could have significant health implications.
The potential therapeutic benefits of Dsup are also being explored in other fields, such as cancer treatment. Simon Galas from the University of Montpellier highlights that low levels of Dsup can extend the lifespan of nematode worms by enhancing their resistance to oxidative stress. This suggests that while Dsup might be beneficial in specific contexts, its effects vary significantly with dosage and application.
In a parallel study, Jessica Tyler at Weill Cornell Medicine has modified yeast to express Dsup at lower levels than those tested by Nislow. Her findings indicate that these lower levels could provide protective benefits without impairing cell growth. Tyler concurs that managing Dsup production is crucial for its future applications.
As research continues, the challenge remains to harness the beneficial properties of Dsup without incurring detrimental effects. Nislow expresses optimism regarding advancements in delivery systems, stating, “There’s so much money and attention on delivery systems. It’s a problem that so many people in pharma are motivated to solve.” This ongoing investigation highlights the intricate balance between innovation and biological complexity as scientists strive to enhance astronaut safety in the cosmos.